 Figure 1. Lower Delmarva Peninsula with the location of Hungars Creek
(Chesapeake Bay side) and Phillips Creek (seaside) indicated.
Figure 1. Lower Delmarva Peninsula with the location of Hungars Creek
(Chesapeake Bay side) and Phillips Creek (seaside) indicated.
Protozoan grazing may be an important link between bacteria and higher trophic levels in aquatic ecosystems (Azam et. al. 1985). In marine aquatic ecosystems, 10 to 50% of the primary production may pass through the free-living, heterotrophic bacteria (Coffin and Sharp 1987). However, it remains unclear whether significant amounts of energy in the form of carbon are cycled through the bacterial community to higher trophic levels or if the primary role of this community is in nutrient regeneration. It is necessary to consider protozoan grazing and its effects on bacterial dynamics to better understand these ecosystem processes.
Bacterial characteristics among bayside and seaside tidal creeks on the Eastern Shore of Virginia have been determined to be significantly different (MacMillian et al. 1992). Abundance, biomass, and productivity are significantly greater in bayside tidal marsh creeks. These observations suggest that there may be differences in the protozoan grazing rates between bayside and seaside tidal marsh creeks. It was therefore, the objective of this study to determine if there remain significant differences in bacterial dynamics among bayside and seaside tidal creeks and if they are correlated with differences in protozoan grazing.
Hungars Creek (bayside) is located on the Chesapeake Bay side of the Delmarva Peninsula. The creek channel is fairly wide in comparison to Phillips Creek (seaside) of the Virginia Coast Reserve and is surrounded by narrow fringes marshes. Phillips Creek is located within the extensive marshes of the barrier island lagoon complex on the eastern side of the Delmarva Peninsula (Fig. 1). The two tidal marsh creeks were sampled approximately every 4-5 weeks beginning in June 1993 and continuing until July 1994.
Protozoan grazing rates were determined using a dilution technique (Landry and Hassett 1982; Tremaine and Mills 1987). Bacterial abundance was measured using the acridine orange direct count method of Hobbie et al. (1984). Bacterial productivity was determined by measuring tritiated thymidine incorporation into bacterial cells (Pollard and Moriarty 1984).
Bacteria in bayside creeks are larger and more abundant than bacteria in seaside creeks even though bacterial doubling times are similar (data not shown). Bacterial carbon was calculated from direct measures of both bacterial abundance and biovolume. A bacterial carbon content of 0.354 pg cell-1 was assumed. The amount of bacterial carbon in bayside creeks is significantly greater than in seaside creeks (Fig. 2). If consumption of the bayside and seaside bacteria by protozoa were similar, then the bayside microbial loop would have the potential to supply a greater amount of carbon and energy to higher trophic levels. However, protozoan grazing constants were significantly greater in seaside than in bayside creeks (data not shown) and the percentage of the seaside bacterial standing stock grazed (Fig. 3) was significantly greater in the bayside suggesting that bacteria are relatively more important as secondary producers in seaside creeks.
 Figure 1. Lower Delmarva Peninsula with the location of Hungars Creek
(Chesapeake Bay side) and Phillips Creek (seaside) indicated.
Figure 1. Lower Delmarva Peninsula with the location of Hungars Creek
(Chesapeake Bay side) and Phillips Creek (seaside) indicated.
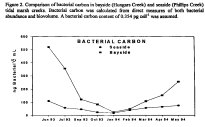 Figure 2. Comparison of bacterial carbon in bayside (Hungars Creek) and seaside
(Phillips Creek) tidal marsh creeks. Bacterial carbon was calculated from
direct measures of both bacterial abundance and biovolume. A bacterial carbon
content of 0.354 pg cell-1 was assumed.
Figure 2. Comparison of bacterial carbon in bayside (Hungars Creek) and seaside
(Phillips Creek) tidal marsh creeks. Bacterial carbon was calculated from
direct measures of both bacterial abundance and biovolume. A bacterial carbon
content of 0.354 pg cell-1 was assumed.
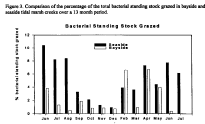 Figure 3. Comparison of the percentage of the total bacterial standing stock
grazed in bayside and seaside tidal marsh creeks over a 13 month period.
Figure 3. Comparison of the percentage of the total bacterial standing stock
grazed in bayside and seaside tidal marsh creeks over a 13 month period.